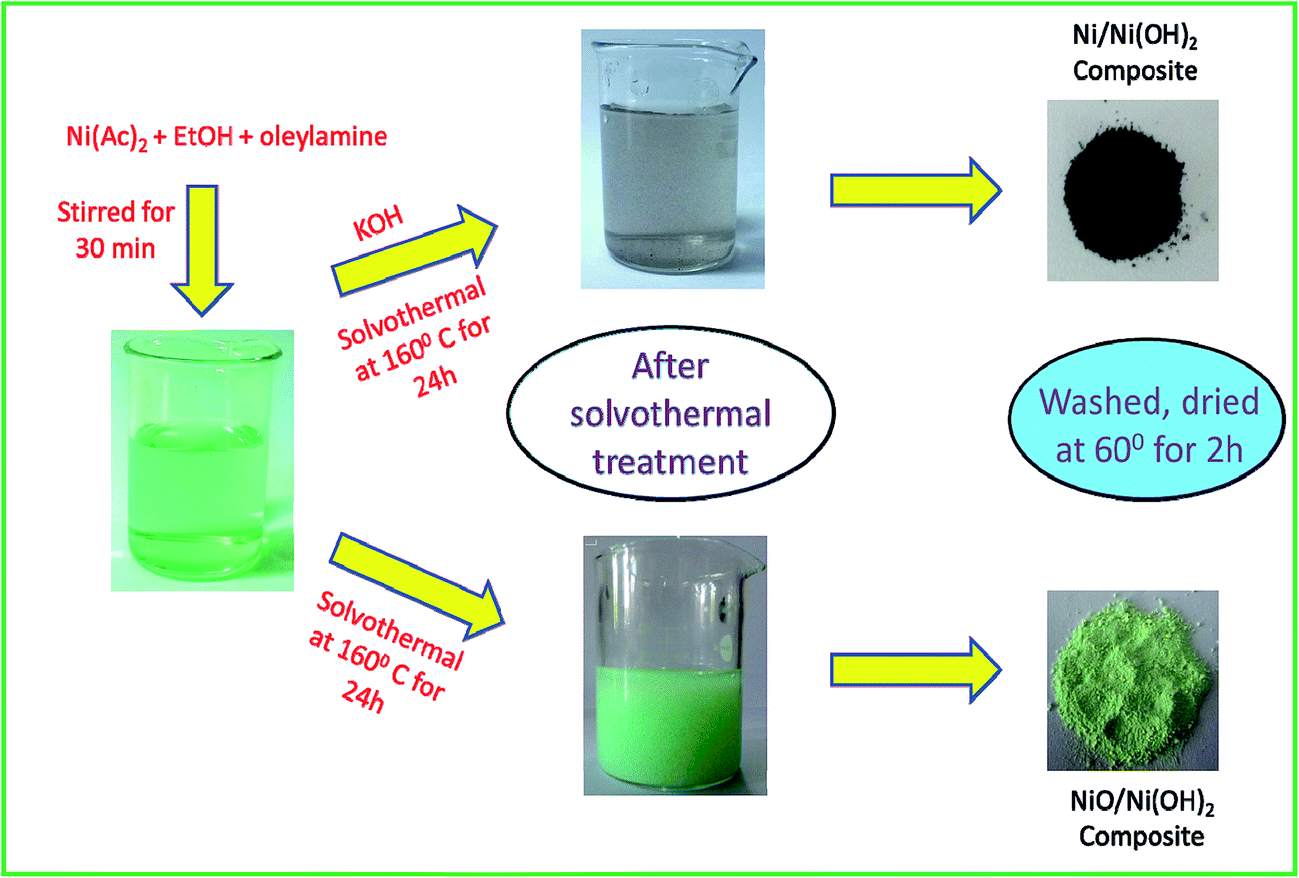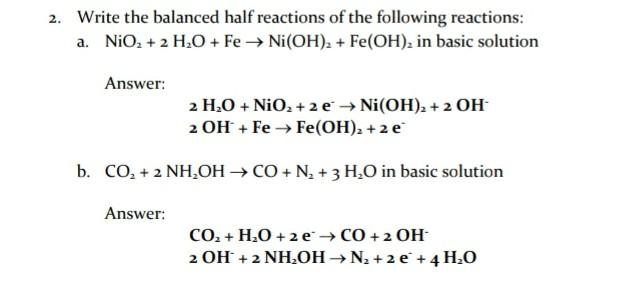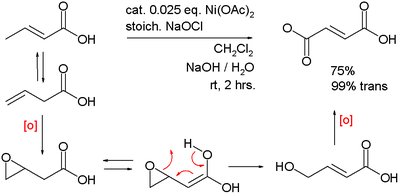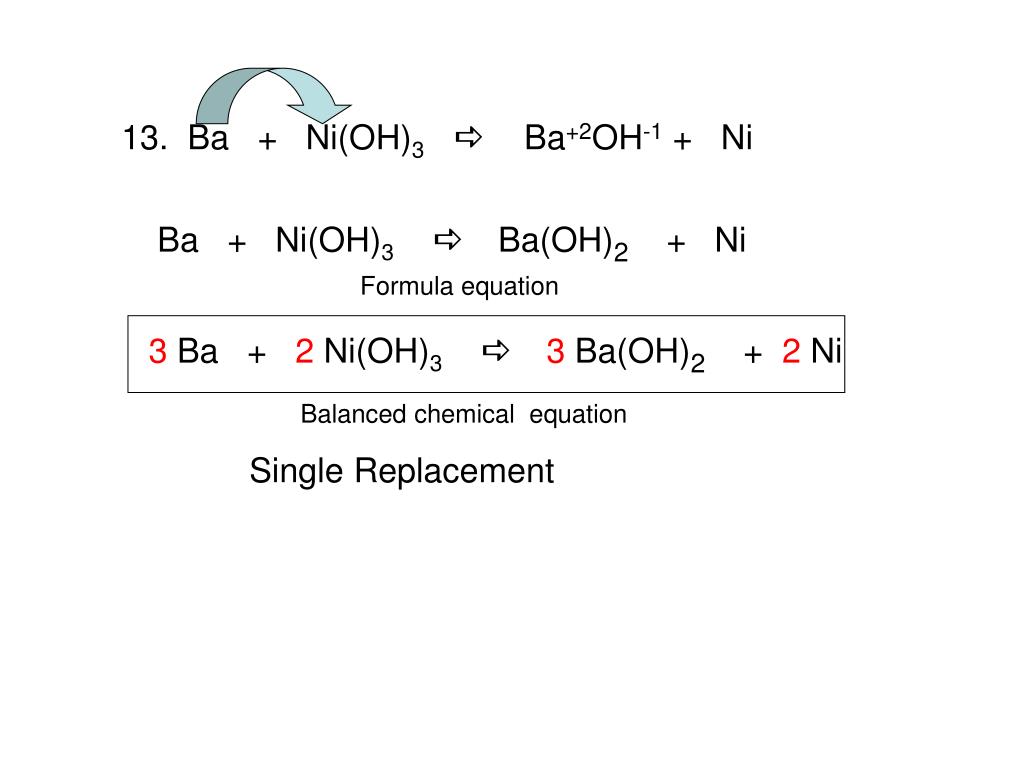
Electrochemical measurements of synthesized nanostructured β-Ni(OH)2 using hydrothermal process and activated carbon based nanoelectroactive materials | SN Applied Sciences

A facile preparation of 3D flower-shaped Ni/Al-LDHs covered by β-Ni(OH)2 nanoplates as superior material for high power application - ScienceDirect

Fabrication of Three-Dimensional Porous NiO/Amorphous Ni(OH)2 Composites for Supercapacitors | Energy & Fuels
![1. Given the following equilibrium constants, calculate the solubility (moles/L) of Ni(OH)2(s) in a solution that has a fixed [OH-] of 3.2x10-7M Ni (OH)2(s) Ksp. - ppt download 1. Given the following equilibrium constants, calculate the solubility (moles/L) of Ni(OH)2(s) in a solution that has a fixed [OH-] of 3.2x10-7M Ni (OH)2(s) Ksp. - ppt download](https://slideplayer.com/15074812/91/images/slide_1.jpg)
1. Given the following equilibrium constants, calculate the solubility (moles/L) of Ni(OH)2(s) in a solution that has a fixed [OH-] of 3.2x10-7M Ni (OH)2(s) Ksp. - ppt download
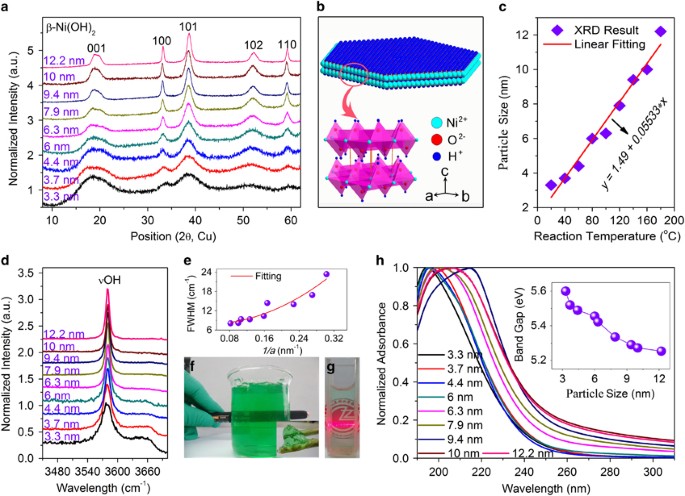
Ultra-small, size-controlled Ni(OH)2 nanoparticles: elucidating the relationship between particle size and electrochemical performance for advanced energy storage devices | NPG Asia Materials
![The Role of the Redox Non‐Innocent Hydroxyl Ligand in the Activation of O2 Performed by [Ni(H)(OH)]+ - Kim - 2023 - Chemistry – A European Journal - Wiley Online Library The Role of the Redox Non‐Innocent Hydroxyl Ligand in the Activation of O2 Performed by [Ni(H)(OH)]+ - Kim - 2023 - Chemistry – A European Journal - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/5a5d2b1b-25f4-4e6d-a83c-c65beeb759da/chem202203128-fig-0003-m.jpg)
The Role of the Redox Non‐Innocent Hydroxyl Ligand in the Activation of O2 Performed by [Ni(H)(OH)]+ - Kim - 2023 - Chemistry – A European Journal - Wiley Online Library
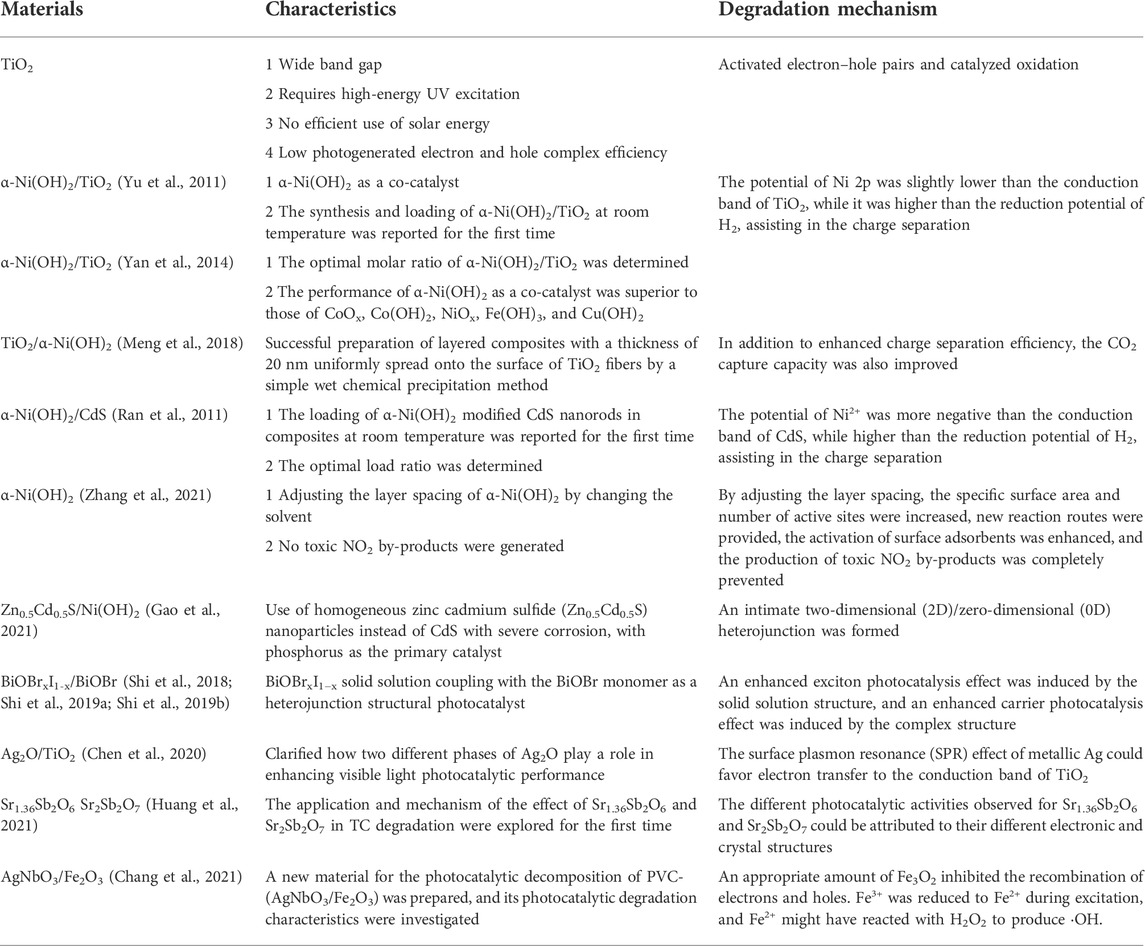
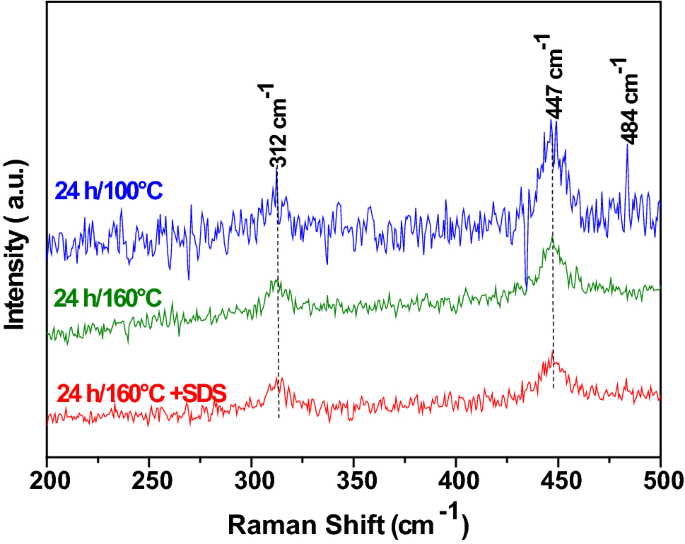
Frontiers | Tunable microscopic aggregation morphology of α-Ni(OH)2 for enhanced photocatalytic degradation of fracturing flowback fluid with ozone synergy

Realizing Two-Electron Transfer in Ni(OH)2 Nanosheets for Energy Storage | Journal of the American Chemical Society
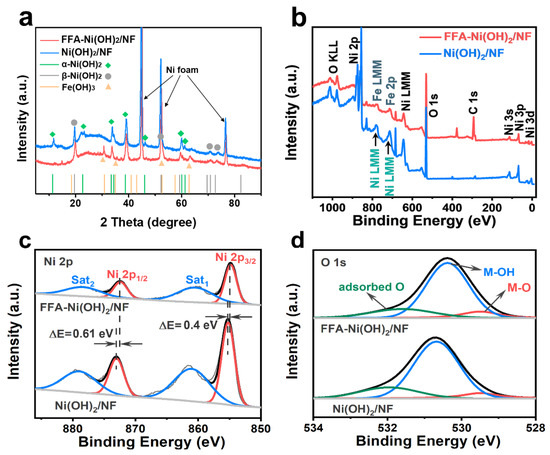
Crystals | Free Full-Text | Ferrocene Formic Acid Surface Modified Ni(OH)2 for Highly Efficient Alkaline Oxygen Evolution

Crystal structures and phase transitions. The crystal structure of (a)... | Download Scientific Diagram

Figure 3 from Electrochemical Performance of β-Nis@Ni(OH)2 Nanocomposite for Water Splitting Applications | Semantic Scholar

Catalysts | Free Full-Text | Catalysts Based on Ni(Mg)Al-Layered Hydroxides Prepared by Mechanical Activation for Furfural Hydrogenation